Source: Tom Williams / Getty
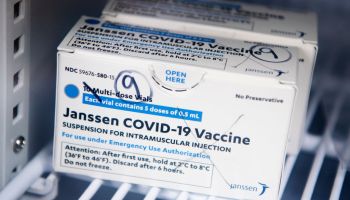
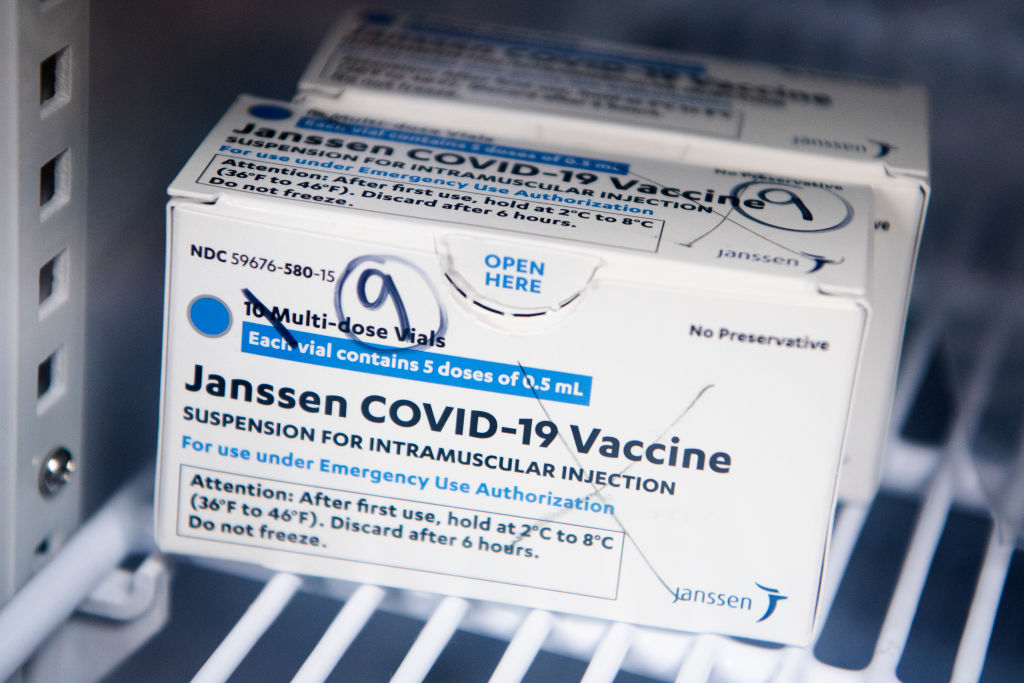
The FDA has announced that the distribution of the Johnson & Johnson COVID-19 will be paused after six women in the U.S. had rare blood-clotting after receiving the vaccine. The women were ranging in ages from 18 to 48, and while six people may not seem like a lot precautions are being taken swiftly. There have been approximately 6.8 million shots given with the majority of people not having issues.
The pausing of the Johnson & Johnson vaccine does not affect the other two vaccines from Pfizer and Moderna. But if you did receive the Johnson & Johnson shot and are having to severe stomach pain, leg cramps, or severe headaches you should call your healthcare provider right away. If you do not know what shot you received, check your CDC vaccination card where it is noted what brand shot you received. If you have an appointment scheduled to receive the Johnson & Johnson COVID-19 vaccine, it is advised to call your provider for further directions.
Get Breaking News & Exclusive Contest in Your Inbox:
The Latest:
- Black Women’s History Month: Korto Momolu Gives Us The Rundown On Her Runway Masterpieces
- Wendy’s: Announces Free FRYdays
- Larsa Pippen & Marcus Jordan: Have Rekindled Romance
- Tyler Perry: Strikes New Deal With BET; 8 Shows Renewed And New Series Ordered
- Regina King Does A Style Switch-Up In Shanghai – And We Are Swooning!
- Is Alabama Killing Prisoners to Steal Their Organs?
- ‘Dancing With the Stars’ Cheryl Burke Reveals She Had Romances With Three Celebrity Partners
- Gov. Ron DeSantis Signs Bill Protecting Florida Police Officers From Civilian Oversight Committees
- Raz-B Reveals The Real Reason B2K Broke Up On TV One’s Upcoming “Uncensored” Episode
- An Exclusive Sneak Peek : Kim Fields Dishes On The Upshaws’ New Season
FDA is Recommending Pausing the Use of Johnson and Johnson Vaccine in the U.S. was originally published on mycolumbusmagic.com